- Home
-
About Us
- Our Founder
- Our Architect
- Genesis & Evolution
- At a Glance
- Vision and Mission
- Core Values
- MoA
- Accreditation & Ranking
- Institutional Development Plan
- Annual Reports
- Mandatory Disclosure
- Policies/Manuals
- Organogram
- Citizen charter
- Institutional Distinctiveness
- Best Practices
- Know Our Institution - Video
- Strategic Plan
- Authorities
- Administration
- Academics
- Research
- Examination
- Schools and Centres
- Facilities
- Student Life
- Placements
- Centres and Cells
- Campus Harmony & Well Being
- Information Corner
- Downloads

Avinashilingam Institute for Home Science and Higher Education for Women
Deemed to be University Estd. u/s 3 of UGC Act 1956, Category A by MHRD [now MoE]
Re-accredited with an 'A++' Grade by NAAC CGPA 3.65/4, Category I by UGC
Coimbatore - 641 043, Tamil Nadu, India


Women Helpline: 181
Child Helpline: 1098
Emergency response support system: 112
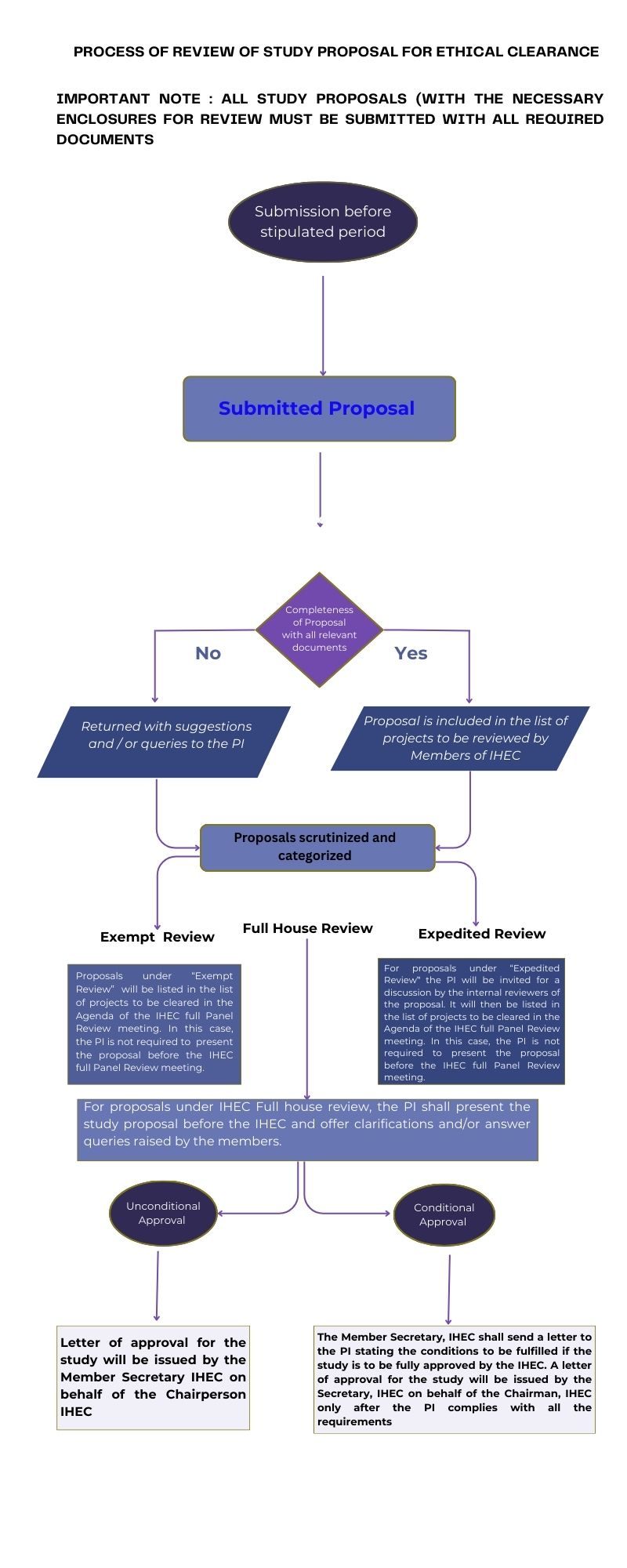
IHEC Application
Documents Required for IHEC Application Download Application Form
Applicants may include all required documents relevant to their study
- Duly filled in application for review by the IHEC (all sections must be complete)
- Declaration form with Signature
Project Timeline/ Gantt Chart showing week-wise activities for the whole study period - Copy of Letter granting funds / Studentship / Scholarship / Fellowship (if applicable)
- Permission letter from the head of the institution from where data is to be collected
- Informed Consent Form in English with due signatures and date. (If study participants are minors, Parental Consent form will be required)
- Informed Consent Form translated into the relevant language(s)
- Patient Information Sheet in English, if applicable
- Patient Information Sheet translated into the relevant language(s), if applicable
- Confidentiality Statement
- Data collection Questionnaire/ Tool in English in English [Enclose sample copy]
- Data collection Questionnaire/ Tool in local language of the respondents [Enclose sample copy]
- Current CV of the Principal Investigator and / or Supervisor
- All relevant pre-clinical animal data
- Compensation for study participation
- Relevant documents for special criteria samples (see page 2)
- Any other information relevant to the study (Provide list)
Additional Documents Required for Special Criteria Study Applications involving studies that involve the following special criteria of study group must include the relevant documents as indicated below:
S.No. |
Special Criteria in Proposal | Additional Documents to be Submitted |
1. | Involves study participants from institutions like schools, orphanages, special needs institutions etc. | Approval letter from the head of the institution |
2. | Involves tribal people (including tribal children) | Approval letter from tribal chief [If study is from a different geographic location other than Coimbatore, a letter from the District Collector should also be enclosed] |
3. | To be executed in a geographic location other than Coimbatore and its suburbs | Approval letter from the IHEC in the area of study execution |
4. | Involves a study group, who do not comprehend English | A translation of the ICF in the local language of the area where the study is being conducted |
5. | Involves any kind of supplementation | Evidence for online registration with CTRI |
6. | Involves any kind of biological sample collection | Acceptance letter from a qualified medical doctor, familiar with the area of research, to act as a member in the study panel (Please ensure that the Registration Number of the medical practitioner is present in the seal or the letter head) |
7. | Any kind of special group as study sample ( e.g. infants,toddlers, very young children, transgenders, mentally compromised individuals (from mild to severe), autistic children, differently- abled individuals, individuals with signs of depression, participants afflicted with sensitive conditions like HIV infection, etc., terminally ill patients, unstable patients, individuals who have suffered second or third degree burns, individuals with multiple medical conditions, etc.) | Acceptance letter from a qualified medical doctor, familiar with the area of research, to act as a member in the study panel (Please ensure that the Registration Number of the medical practitioner is present in the seal or the letter head) |
IHEC Fee details
Fees for Review of Applications for Institutional Human Ethical Clearance
2025-2026
Applicants submitting applications for review for obtaining Human Ethical Clearance may pay the fees at the finance section in-person. Online payment may also be made.
Fee Details
Level of Study Programme | Fees to be Paid (Rs) |
For In-Campus Applicants | |
PG Students | 200 |
Ph. D Scholars | 500 |
Mini projects (Single Investigator) | 1200 |
MRD/Major Projects (Single Investigator) | 2000 |
For Applicants fromOutside Institutions | |
PG Students | 1000 |
Ph. D Scholars | 1200 |
Mini projects (Single Investigator) | 1500 |
MRD/Major Projects (Single Investigator) | 2000 |
IHEC Instructions to Investigators
Instructions to Investigators/Applicants for IHEC Review
Instructions to Research Supervisors/Project Investigators/Applicants for IHEC Review
Research Supervisors, Ph.D scholars and students involved in thesis project work and Minor and Major Projects may submit their study proposal applications for IHEC review by second week of September/January/May every year.
The application format with all necessary enclosures is available in our institution website: https://avinuty.ac.in/research-committees/research-advisory-committees
Prepare your application for IHEC review as per the format provided
Register details required in the Google form at the link below: https://tinyurl.com/IHECregistration
Soft copy of the proposal in PDF format and fee receipt should be attached in the Google form
One print copy of the complete application with copy of fee receipt must be submitted to the member-secretary
Applications for IHEC review may be submitted within the second week of September/January/May every year
Queries may be addressed to the member secretary at ihec@avinuty.ac.in with the Name of PI and department name in the subject line. Kindly note that for all research studies including sponsored projects and institutional minor projects involving human participants and collection of human data, Institutional Human Ethics Committee clearance is compulsory
Important Note:
1. Research work/phase of study alone may be granted Ethical clearance. The title of the research work mentioned at this stage is provisional and subject to revision at the time of synopsis submission. The final title of the PhD thesis may therefore differ from the current working title. However the ethical clearance has been duly obtained for the proposed research work. Any change in the thesis title will not involve a change in the scope of study for which the ethical clearance was granted
2. Validity of the IHEC certificate The IHEC certificate is exclusively for the particular study/project and for the particular study period and not for the follow up and after study period 3. Any amendment/inclusion and exclusion of samples, extension of previous research work with ethical clearance by other scholars maybe considered as a new submission only.
IHEC guidelines
Guidelines
ICMR National Ethical Guidelines (India) 2017 ... Click to view
New Drugs and Clinical Trials Rules (2019, India) ... Click to view
- Home
-
About Us
- Our Founder
- Our Architect
- Genesis & Evolution
- At a Glance
- Vision and Mission
- Core Values
- MoA
- Accreditation & Ranking
- Institutional Development Plan
- Annual Reports
- Mandatory Disclosure
- Policies/Manuals
- Organogram
- Citizen charter
- Institutional Distinctiveness
- Best Practices
- Know Our Institution - Video
- Strategic Plan
- Authorities
- Administration
- Academics
- Research
- Examination
- Schools and Centres
- Facilities
- Student Life
- Placements
- Centres and Cells
- Campus Harmony & Well Being
- Information Corner
- Downloads